Rezdiffra (resmetirom) has been approved by the FDA of the USA for the treatment of adults with noncirrhotic non-alcoholic steatohepatitis (NASH) with moderate to advanced liver scarring (fibrosis), to be used along with diet and exercise.
Until now, patients with noncirrhotic non-alcoholic steatohepatitis (NASH) who also have notable liver scarring did not have a medication that could directly address their liver damage. FDA’s approval of Rezdiffra will, for the first time, provide a treatment option for these patients, in addition to diet and exercise.
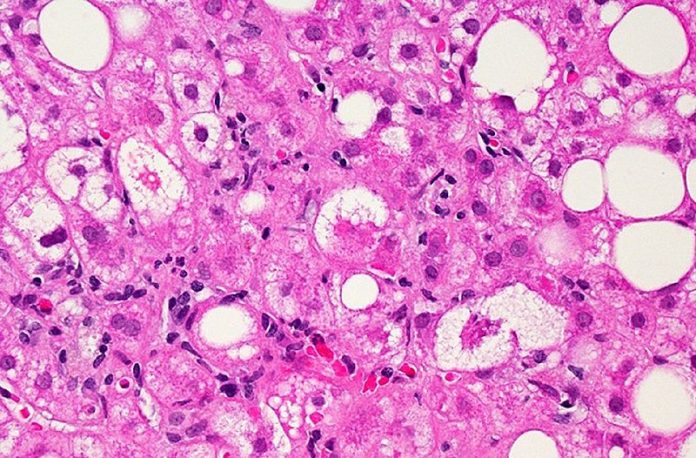
NASH is a result of the progression of non-alcoholic fatty liver disease where liver inflammation, over time, can lead to liver scarring and liver dysfunction. NASH is often associated with other health problems such as high blood pressure and type 2 diabetes. By at least one estimate, approximately 6-8 million people in the U.S. have NASH with moderate to advanced liver scarring, with that number expected to increase.
Rezdiffra is a partial activator of a thyroid hormone receptor; activation of this receptor by Rezdiffra in the liver reduces liver fat accumulation.
Safety and efficacy of Rezdiffra
The safety and efficacy of Rezdiffra was evaluated based on an analysis of a surrogate endpoint at month 12 in a 54-month, randomized, double-blind placebo-controlled trial. The surrogate endpoint measured the extent of liver inflammation and scarring. The sponsor is required to conduct a postapproval study to verify and describe Rezdiffra’s clinical benefit, which will be done through completing the same 54-month study, which is still ongoing. To enroll in the trial, patients needed to have a liver biopsy showing inflammation due to NASH with moderate or advanced liver scarring. In the trial, 888 subjects were randomly assigned to receive one of the following: placebo (294 subjects); 80 milligrams of Rezdiffra (298 subjects); or 100 milligrams of Rezdiffra (296 subjects); once daily, in addition to standard care for NASH, which includes counseling for healthy diet and exercise.
At 12 months, liver biopsies showed that a greater proportion of subjects who were treated with Rezdiffra achieved NASH resolution or an improvement in liver scarring as compared with those who received the placebo. A total of 26% to 27% of subjects who received 80 milligrams of Rezdiffra and 24% to 36% of subjects who received 100 milligrams of Rezdiffra experienced NASH resolution and no worsening of liver scarring, compared to 9% to 13% of those who received placebo and counseling on diet and exercise. The range of responses reflects different pathologists’ readings. In addition, a total of 23% of subjects who received 80 milligrams of Rezdiffra and 24% to 28% of subjects who received 100 milligrams of Rezdiffra experienced an improvement in liver scarring and no worsening of NASH, compared to 13% to 15% of those who received placebo, depending on each pathologist’s readings. Demonstration of these changes in a proportion of patients after just one year of treatment is notable, as the disease typically progresses slowly with most patients taking years or even decades to show progression.
Side effects of Rezdiffra
The most common side effects of Rezdiffra included diarrhea and nausea. Rezdiffra comes with certain warnings and precautions, such as drug-induced liver toxicity and gallbladder-related side effects.
Use of Rezdiffra should be avoided in patients with decompensated cirrhosis. Patients should stop using Rezdiffra if they develop signs or symptoms of worsening liver function while on Rezdiffra treatment.
Drug interactions of Rezdiffra
Using Rezdiffra at the same time as certain other drugs, in particular statins for lowering cholesterol, may result in potentially significant drug interactions. Health care providers should refer to the full prescribing information for additional information on these potentially significant drug interactions with Rezdiffra, recommended dosage and administration modifications.
The FDA approved Rezdiffra under the accelerated approval pathway, which allows for earlier approval of drugs that treat serious conditions and address an unmet medical need, based on a surrogate or intermediate clinical endpoint that is reasonably likely to predict clinical benefit. The required aforementioned 54-month study, which is ongoing, will assess clinical benefit after 54 months of Rezdiffra treatment.
Rezdiffra received Breakthrough Therapy, Fast Track and Priority Review designations for this indication.
The FDA granted the approval of Rezdiffra to Madrigal Pharmaceuticals.
***
Source:
FDA 2024. News release – FDA Approves First Treatment for Patients with Liver Scarring Due to Fatty Liver Disease. Posted 14 March 2024. Available at https://www.fda.gov/news-events/press-announcements/fda-approves-first-treatment-patients-liver-scarring-due-fatty-liver-disease
***